
Loading, please wait...

Loading, please wait...
"Wherever the art of Medicine is loved, there is also a love of Humanity."
— Hippocrates
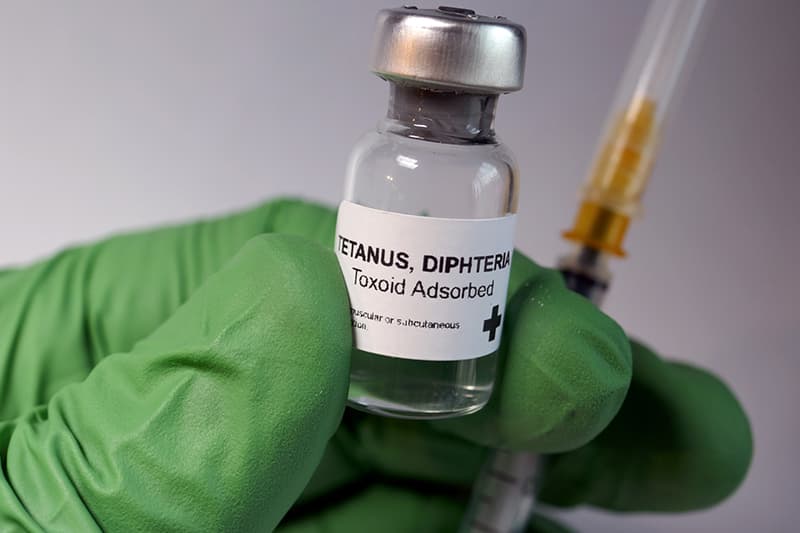
Union Health Minister JP Nadda recently marked a significant healthcare milestone with the Td vaccine launch India. This initiative replaces the traditional Tetanus Toxoid (TT) vaccine within the Universal Immunization Programme (UIP). Consequently, the move aligns India with global standards set by the World Health Organization (WHO). The Central Research Institute (CRI) in Kasauli will spearhead the domestic production of this vital vaccine. Moreover, this transition ensures better protection against diphtheria among adolescents and adults across the country.
Extensive scientific evidence indicates that childhood immunization significantly reduces the incidence of diphtheria and tetanus. However, antibody levels often decline over time, especially regarding diphtheria protection. Therefore, booster doses become necessary to maintain immunity. Since 2006, the WHO has recommended that countries transition from TT to the Td vaccine. This recommendation received further validation through the 2017 WHO Tetanus Vaccine Position Paper. Because of these global insights, India decided to upgrade its immunization protocol.
The transition to the Td vaccine aims to strengthen protection against diphtheria while sustaining gains in tetanus elimination. Specifically, the vaccine combines purified diphtheria toxoid and purified tetanus toxoid with a reduced antigen content for the diphtheria component. This formulation provides robust immunity for adults without increasing the risk of adverse reactions. Additionally, the National Technical Advisory Group on Immunization (NTAGI) recommended this replacement for all age groups. This includes pregnant women, who play a critical role in maternal and neonatal tetanus prevention.
Furthermore, the domestic manufacturing of the Td vaccine by CRI Kasauli underscores India's self-reliance in vaccine production. The Institute successfully navigated developmental studies and secured necessary licenses for commercial manufacturing. Subsequently, CRI plans to supply 55 lakh doses to the UIP by April 2026. This domestic supply chain will ensure the sustained availability of quality-assured vaccines. Consequently, healthcare providers can now offer broader protection against two potentially life-threatening infections through a single injection.
Tetanus remains a serious disease characterized by painful muscle stiffness and spasms. Often called lockjaw, it can lead to severe complications or even death if left untreated. Similarly, diphtheria is a life-threatening infection that affects the respiratory system and heart. It can cause breathing difficulties, paralysis, and heart failure. By introducing the Td vaccine, the government aims to reduce the morbidity and mortality associated with these vaccine-preventable diseases. This proactive step reflects the ongoing commitment to public health and disease eradication in India.
Q1: Why is the Td vaccine replacing the TT vaccine in India?
The Td vaccine replaces the TT vaccine because it provides protection against both tetanus and diphtheria. While TT only protects against tetanus, Td addresses the declining immunity against diphtheria in adolescents and adults.
Q2: Who should receive the Td vaccine under the new guidelines?
According to the National Immunization Programme, the Td vaccine is recommended for all age groups previously receiving TT. This includes adolescents, adults, and pregnant women to ensure comprehensive protection.
Q3: Is the Td vaccine safe for pregnant women?
Yes, health authorities like NTAGI have approved the Td vaccine for use in pregnant women. It helps prevent maternal and neonatal tetanus while also providing protection against diphtheria.
Disclaimer: This content is for informational and educational purposes only. It does not constitute medical advice or replace professional judgment. Refer to the latest local and national guidelines for clinical practice.
References

Union Health Minister JP Nadda launches the Td vaccine at CRI Kasauli, replacing TT to boost diphtheria and tetanus protection across India's immunization p...
3 months ago

Stanford scientists cured Type 1 diabetes in mice by transplanting stem and islet cells under a gentle, low-radiation protocol to reset the immune system....
Today

The SRS 2024 report highlights a decline in India's infant mortality rate to 24, though severe state-level disparities and neonatal care gaps persist....
Today

New registry data shows that a lower estimated glucose disposal rate (eGDR), a proxy for insulin resistance, significantly increases the risk of first MI....
Today

Researchers find that FLNB and TTC26 regulate ciliary Hedgehog signaling to maintain intervertebral disc matrix and prevent adolescent idiopathic scoliosis....
Today

Semaglutide significantly reduces body weight and HbA1c in patients with schizophrenia spectrum disorders, according to a 2026 systematic review and meta-an...
Today